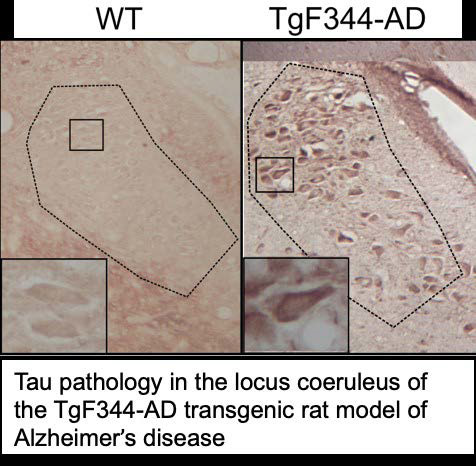
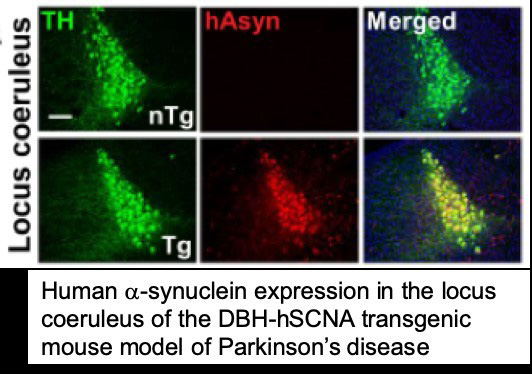
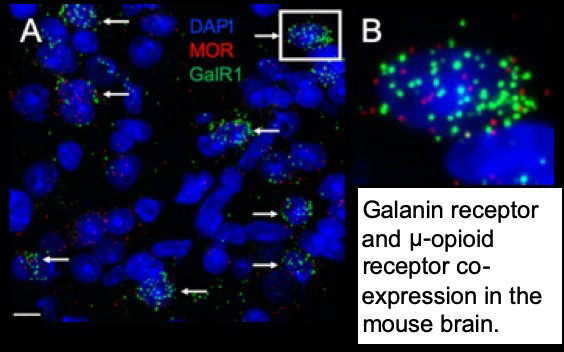
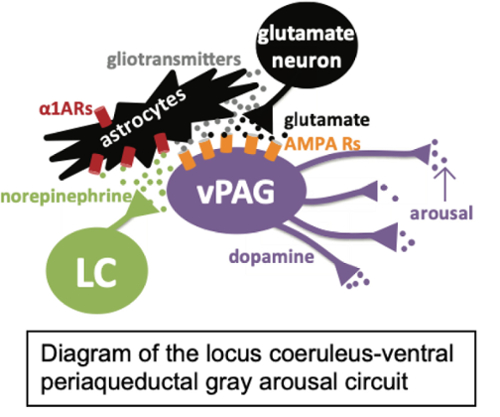
Lab Overview
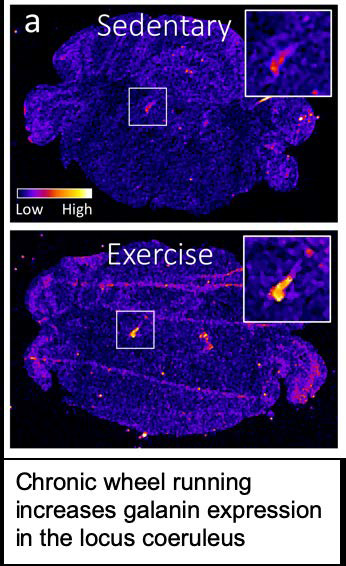
My approach is to use a combination of genetics, behavior, pharmacology, neurochemistry, optogenetics, and chemogenetics in rodents to better understand the neurobiology underlying normal brain function as well as neurological and neuropsychiatric disease. These include Alzheimer’s disease, Parkinson’s disease, drug addiction, sleep disorders, depression, and anxiety. Most projects in the lab focus on a brainstem region called the locus coeruleus (LC), the major norepinephrine (NE) nucleus in the brain. LC neurons project to almost every region of the brain; in fact, it is nearly impossible to find a brain structure that completely lacks noradrenergic input. The brain noradrenergic system is critical for arousal, attention, stress responses, and certain aspects of learning and memory. Although LC neurons use NE as their primary neurotransmitter, they also synthesize and release several other co-transmitters. These include dopamine, which is actually a biosynthetic precursor for NE, as well as neuropeptides such as galanin, neuropeptide Y (NPY), and brain-derived neurotrophic factor (BDNF). A major goal of our research program is to understand how LC co-transmission is regulated and how the signals from these various neuromodulators are integrated and interpreted by LC target cells.
Lab Faculty

David Weinshenker, PhD
Professor, Department of Human Genetics
Scientific Director, Rodent Behavioral Core
Assistant Dean and Director, Office of Postdoctoral and Mentored Trainee Education
Director, Research Education Component, Goizueta Alzheimer’s Disease Research Center
Co-PI, AD Big Data to Biology Training Program T32